Robohub.org
Molecular Robotics at the Wyss Institute
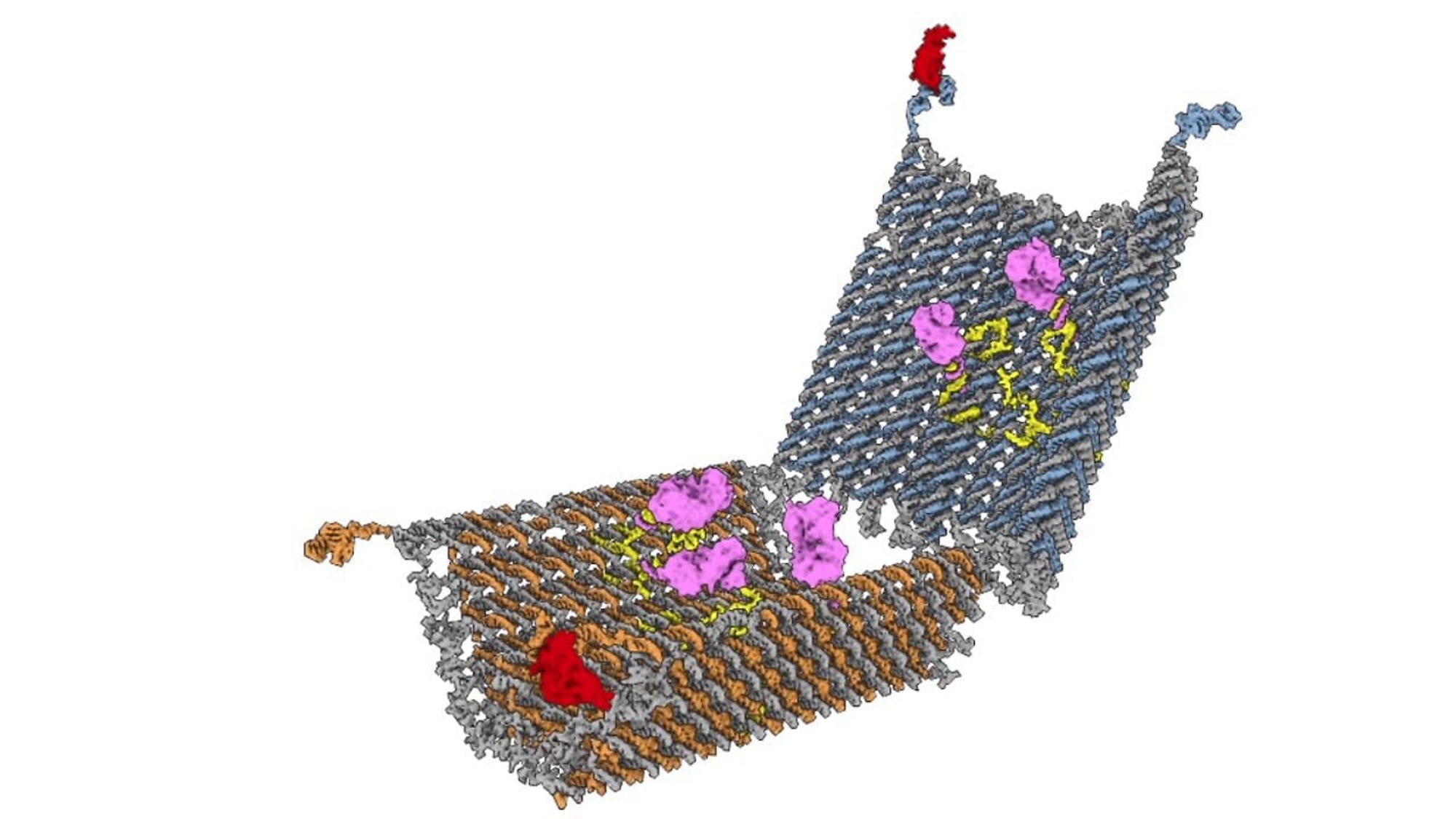
This programmable DNA nanorobot ‘patrols’ the bloodstream and releases its payload of drugs in response to the presence of its target, much like the body’s white blood cells. Credit: Wyss Institute at Harvard University
By Lindsay Brownell
DNA has often been compared to an instruction book that contains the information needed for a living organism to function, its genes made up of distinct sequences of the nucleotides A, G, C, and T echoing the way that words are composed of different arrangements of the letters of the alphabet. DNA, however, has several advantages over books as an information-carrying medium, one of which is especially profound: based on its nucleotide sequence alone, single-stranded DNA can self-assemble, or bind to complementary nucleotides to form a complete double-stranded helix, without human intervention. That would be like printing the instructions for making a book onto loose pieces of paper, putting them into a box with glue and cardboard, and watching them spontaneously come together to create a book with all the pages in the right order.
But just as paper can also be used to make origami animals, cups, and even the walls of houses, DNA is not limited to its traditional purpose as a passive repository of genetic blueprints from which proteins are made – it can be formed into different shapes that serve different functions, simply by controlling the order of As, Gs, Cs, and Ts along its length. A group of scientists at the Wyss Institute for Biologically Inspired Engineering at Harvard University is investigating this exciting property of DNA molecules, asking, “What types of systems and structures can we build with them?”
They’ve decided to build robots.
At first glance, there might not seem to be much similarity between a strand of DNA and, say, a Roomba™ or Rosie the Robot from The Jetsons. “Looking at DNA versus a modern-day robot is like comparing a piece of string to a tractor trailer,” says Wyss Faculty member Wesley Wong, Ph.D., Assistant Professor of Biological Chemistry and Molecular Pharmacology (BCMP) at Harvard Medical School (HMS) and Investigator at Boston Children’s Hospital. Despite the vast difference in their physical form, however, robots and DNA share the ability to be programmed to complete a specific function – robots with binary computer code, DNA molecules with their nucleotide sequences.
Recognizing that commonality, the Wyss Institute created the cross-disciplinary Molecular Robotics Initiative in 2016, which brings together researchers with experience in the disparate disciplines of robotics, molecular biology, and nanotechnology to collaborate and help inform each other’s work to solve the fields’ similar challenges. Wong is a founding member of the Initiative, along with Wyss Faculty members William Shih, Ph.D., Professor of BCMP at HMS and Dana-Farber Cancer Institute; Peng Yin, Ph.D., Professor of Systems Biology at HMS; and Radhika Nagpal, Ph.D., Fred Kavli Professor of Computer Science at Harvard’s John A. Paulson School of Engineering and Applied Sciences (SEAS); as well as other Wyss scientists and support staff.
“We’re not used to thinking about molecules inside cells doing the same things that computers do. But they’re taking input from their environment and performing actions in response – a gene is either turned on or off, a protein channel is either open or closed, etc. – in ways that can resemble what computer-controlled systems do,” says Shih. “Molecules can do a lot of things on their own that robots usually have trouble with (move autonomously, self-assemble, react to the environment, etc.), and they do it all without needing motors or an external power supply,” adds Wyss Founding Director Don Ingber, M.D., Ph.D., who is also the Judah Folkman Professor of Vascular Biology at HMS and the Vascular Biology Program at Boston Children’s Hospital, as well as a Professor of Bioengineering at SEAS. “Programmable biological molecules like DNA have almost limitless potential for creating transformative nanoscale devices and systems.”

Molecular Robotics capitalizes on the recent explosion of technologies that read, edit, and write DNA (like next-generation sequencing and CRISPR) to investigate the physical properties of DNA and its single-stranded cousin RNA. “We essentially treat DNA not only as a genetic material, but as an incredible building block for creating molecular sensors, structures, computers, and actuators that can interact with biology or operate completely separately,” says Tom Schaus, M.D., Ph.D., a Staff Scientist at the Wyss Institute and Molecular Robotics team member.
Many of the early projects taking advantage of DNA-based self-assembly were static structures. These include DNA “clamshell” containers that can be programmed to snap open and release their contents in response to specific triggers, and DNA “bricks” whose nucleotide sequences allow their spontaneous assembly into three-dimensional shapes, like tiny Lego™ bricks that put themselves together to create sculptures automatically. Many of these structures are three-dimensional, and some incorporate as many as 10,000 unique DNA strands in a single complete structure.
The reliable specificity of DNA and RNA (where A always binds with T or U, C always with G) allows for not only the construction of static structures, but also the programming of dynamic systems that sense and respond to environmental cues, as seen in traditional robotics. For example, Molecular Robotics scientists have created a novel, highly controllable mechanism that automatically builds new DNA sequences from a mixture of short fragments in vitro. It utilizes a set of hairpin-shaped, covalently-modified DNA strands with a single-stranded “overhang” sequence dangling off one end of the hairpin. The overhang sequence can bind to a complementary free-floating fragment of DNA (a “primer”) and act as a template for its extension into a double-stranded DNA sequence. The hairpin ejects the new double strand and can then be re-used in subsequent reactions to produce multiple copies of the new strand.

Such extension reactions can be programmed to occur only in the presence of signal molecules, such as specific RNA sequences, and can be linked together to create longer DNA product strands through “Primer Exchange Reactions” (PER). PER can in turn be programmed to enzymatically cut and destroy particular RNA sequences, record the order in which certain biochemical events happen, or generate components for DNA structure assembly.
PER reactions can also be combined into a mechanism called “Autocycling Proximity Recording” (APR), which records the geometry of nano-scale structures in the language of DNA. In this instance, unique DNA hairpins are attached to different target molecules in close proximity and, if any two targets are close enough together, produce new pieces of DNA containing the molecular identities (“names”) of those two targets, allowing the shape of the underlying structure to be determined by sequencing that novel DNA.
Another tool, called “toehold switches,” can be used to exert complex and precise control over the machinery inside living cells. Here, a different, RNA-based hairpin is designed to “open” when it binds to a specific RNA molecule, exposing a gene sequence in its interior that can be translated into a protein that then performs some function within the cell. These synthetic circuits can even be built with logic-based sequences that mimic the “AND,” “OR,” and “NOT” system upon which computer languages are based, which prevents the hairpin from opening and its gene from being translated except under very specific conditions.
Such an approach could induce cells that are deficient in a given protein to produce more of it, or serve as a synthetic immune system that, when it detects a given problem in the cell, produces a toxin that kills it to prevent it from spreading an infection or becoming cancerous. [toeholds] “Because we have a thorough understanding of DNA and RNA’s properties and how their bases pair together, we can use that simple machinery to design complex circuits that allow us to precisely interact with the molecular world,” says Yin. “It’s an ability that has been dreamed about for a long time, and now, we’re actually making it a reality.”
The potential applications of that ability are seemingly endless. In addition to the previously mentioned tools, Molecular Robotics researchers have created loops of DNA attached to microscopic beads to create “calipers” that can both measure the size, structure, and stiffness of other molecules, and form the basis of inexpensive protein recognition tests. Another advance is folding single-stranded DNA into molecular origami to create molecular structures, rather than traditional double-stranded DNA. Some academic projects are already moving into the commercial sector. These include a low-cost alternative to super-resolution microscopy that can image up to 100 different molecular targets in a single sample (DNApaint), as well as a multiplexed imaging technique that integrates fluorescent probes into self-folding DNA structures and enables simultaneous visualization of ultra-rare DNA and/or RNA molecules.
We’re trying to push the limits of these really dumb little molecules to get them to behave in sophisticated, collective ways – it’s a new frontier for DNA nanotechnology.
One of the major benefits of engineering molecular machines is that they’re tiny, so it’s relatively easy to create a large amount of them to complete any one task (for example, circulating through the body to detect any rogue cancer DNA). Getting simple, individual molecules to interact with each other to achieve a more complex, collective task (like relaying the information that cancer has been found), however, is a significant challenge, and one that the roboticists in Molecular Robotics are tackling at the macroscopic scale with inch-long “Kilobots.”
Taking cues from colonies of insects like ants and bees, Wyss researchers are developing swarms of robots that are themselves limited in function but can form complex shapes and complete tasks by communicating with each other via reflected infrared light. The insights gained from studies with the Kilobots are likely to be similar to those needed to solve similar problems when trying to coordinate molecular robots made of DNA.

“In swarm robotics, you have multiple robots that explore their environment on their own, talk to each other about what they find, and then come to a collective conclusion. We’re trying to replicate that with DNA but it’s challenging because, as simple as Kilobots are, they’re brilliant compared to DNA in terms of computational power,” says Justin Werfel, Ph.D., a Senior Research Scientist at the Wyss Institute and director of the Designing Emergence Laboratory at Harvard. “We’re trying to push the limits of these really dumb little molecules to get them to behave in sophisticated, collective ways – it’s a new frontier for DNA nanotechnology.”
Given the magnitude of the challenge and the short time the Molecular Robotics Initiative has existed, it is already making significant progress, with more than two dozen papers published and two companies (Ultivue and NuProbe) founded around its insights and discoveries. It may take years of creative thinking, risk taking, and swapping ideas across the members’ different expertise areas before a molecule of DNA is able to achieve the same task on the nanoscale that a robot can do on the human scale, but the team is determined to see it happen.
“Our vision with Molecular Robotics is to solve hard problems humanity currently faces using smaller, simpler tools, like a single loop of DNA or a single Kilobot that can act cooperatively en masse, instead of bigger, more complex ones that are harder to develop and become useless should any one part fail,” says Wong. “It’s an idea that definitely goes against the current status quo, and we’re lucky enough to be pursuing it here at the Wyss Institute, which brings together people with common goals and interests to create new things that wouldn’t exist otherwise.”
Click on the links below to explore research from the Molecular Robotics Initiative.
- Researchers at Harvard’s Wyss Institute Develop DNA Nanorobot to Trigger Targeted Therapeutic Responses
- A 100-fold leap to GigaDalton DNA nanotech
- Autonomously growing synthetic DNA strands
- High-fidelity recording of molecular geometry with DNA “nanoscopy”
- Programming cells with computer-like logic
- Democratizing high-throughput single molecule force analysis
- Single-stranded DNA and RNA origami go live
- Capturing ultrasharp images of multiple cell components at once
- A self-organizing thousand-robot swarm
- Discrete Molecular Imaging